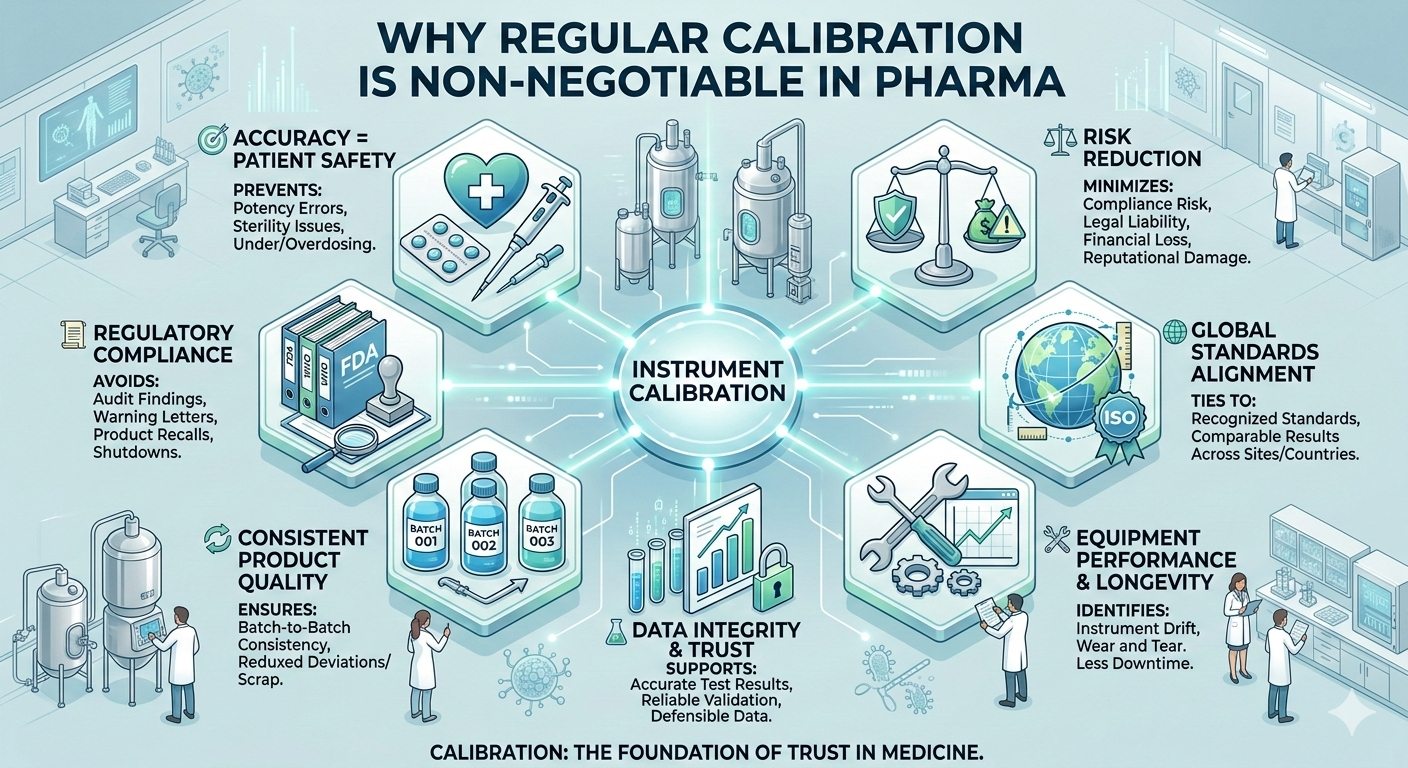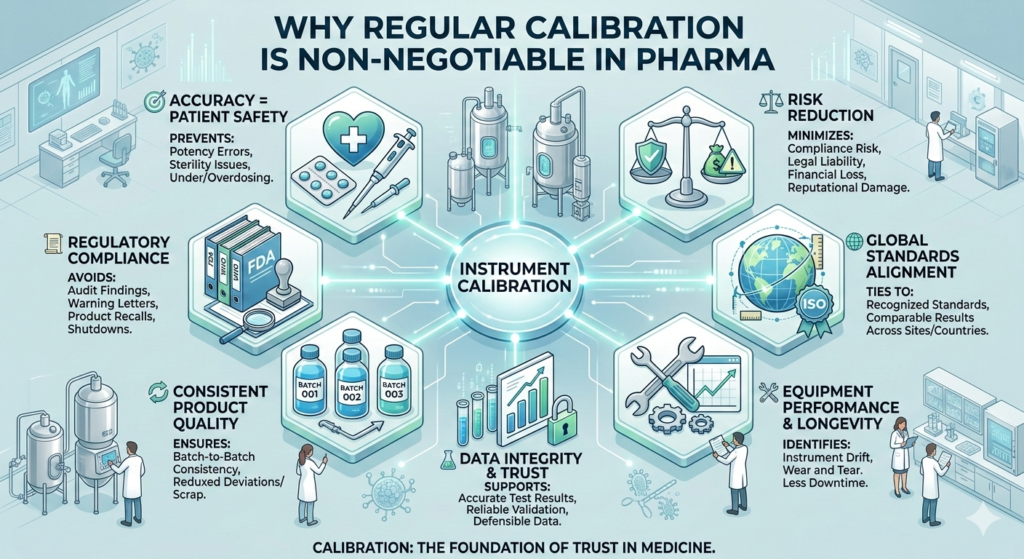
In the pharmaceutical industry, precision is not optional. Every process, from raw material testing to final product release, relies on accurate measurements. Regular calibration of equipment ensures that these measurements remain reliable, compliant, and safe for patients.
Ensuring Patient Safety
Pharmaceutical instruments measure critical parameters such as temperature, pressure, weight, pH, and volume. Even minor inaccuracies can affect drug strength, stability, or sterility. Regular calibration ensures that equipment performs within defined limits, helping prevent dosing errors and protecting patient health.
Meeting Regulatory Requirements
Pharma manufacturers must comply with strict regulatory standards set by authorities such as the FDA and the WHO. These regulations require documented evidence that equipment is calibrated and traceable to recognized standards. Failure to maintain calibration programs can result in audit observations, warning letters, recalls, or production delays.
Maintaining Product Quality and Consistency
Consistent product quality depends on consistent measurements. Equipment drift over time can lead to batch variations, deviations, or failed quality tests. Regular calibration helps ensure uniformity across batches, reducing waste and improving overall process reliability.
Supporting Data Integrity
Pharmaceutical decisions are driven by data. Calibration plays a vital role in ensuring the accuracy and credibility of laboratory and manufacturing data. Reliable instruments support valid test results, strong validation studies, and trustworthy records—key elements of data integrity and regulatory compliance.
Improving Equipment Performance and Reliability
Calibration helps identify equipment drift, wear, or malfunction before it leads to failure. This proactive approach reduces unplanned downtime, extends equipment life, and supports smooth production operations.
Alignment with Global Quality Standards
Calibration ensures measurement traceability to internationally accepted standards such as those defined by the ISO. This is especially important for companies operating multiple facilities or working with global partners, as it ensures consistency across locations.
Reducing Business and Compliance Risk
Inaccurate measurements can lead to product recalls, regulatory action, financial loss, and reputational damage. A robust calibration program minimizes these risks by ensuring ongoing compliance and operational control.